What Is Plasmapheresis? Benefits, Risks, and Treatment Outcomes Explained
When harmful antibodies or inflammatory molecules circulate through the bloodstream, they can trigger devastating effects on nerves, blood vessels, and organs. Plasmapheresis - sometimes called therapeutic plasma exchange or TPE - offers a way to physically extract these damaging substances from the body. For decades, this procedure has served as a cornerstone treatment for conditions ranging from rare blood disorders to debilitating neurological diseases. Understanding how plasmapheresis works, which conditions respond to it, and what the research evidence actually shows can help patients and families navigate treatment decisions alongside their medical teams.
[banner2]
What Is Plasmapheresis?
At its core, plasmapheresis involves separating blood into its component parts, removing the liquid plasma fraction, and returning the cellular elements back to the patient with fresh replacement fluid. The name itself combines the Greek root "plasma" (meaning something molded or formed) with "apheresis" (meaning removal or taking away).
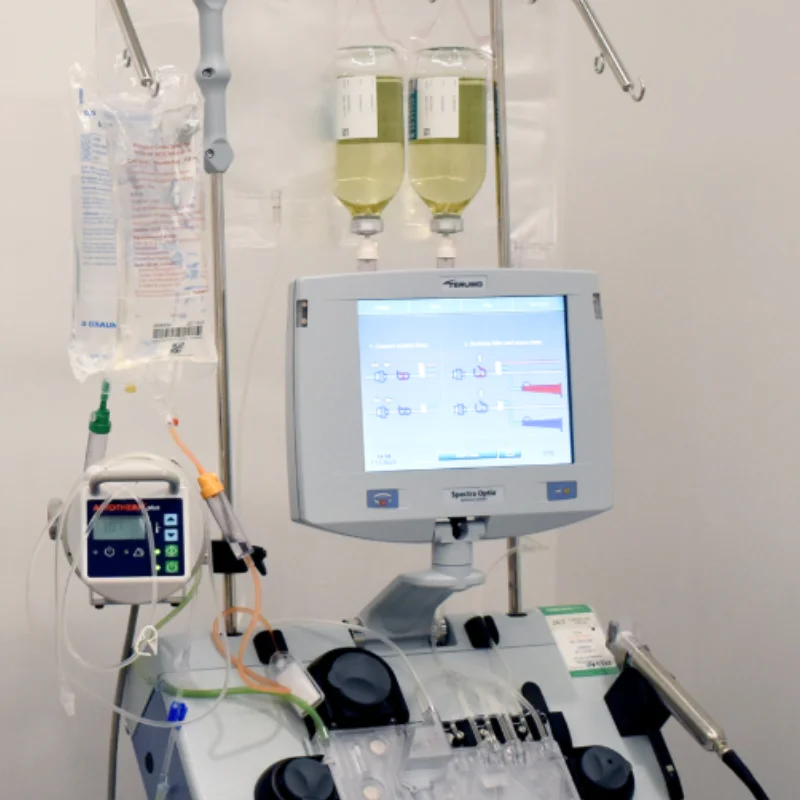
The Mechanics Behind Plasma Separation
Modern plasmapheresis machines accomplish this separation through one of two technical approaches (StatPearls, 2024):
Centrifugal separation spins the blood at high speed, causing components to layer based on their density. Red cells, being heaviest, settle to the outside, while plasma - the lightest fraction - concentrates toward the center where it can be selectively removed.
Membrane-based filtration passes blood through specialized filters with pores sized to allow plasma through while retaining larger cellular elements. This approach resembles the kidney's natural filtration mechanism but targets different substances.
Regardless of which method is employed, the extracted plasma - potentially laden with pathogenic autoantibodies, inflammatory cytokines, immune complexes, and toxic metabolites - is discarded. The patient receives replacement fluid, typically human albumin solution or, when coagulation factors must be replenished, fresh frozen plasma (Nguyen et al., American Journal of Kidney Diseases, 2023).
Clinical studies indicate that each plasmapheresis session removes roughly 60% of circulating coagulation factors and approximately 85% of fibrinogen when albumin serves as the replacement solution. Beyond simple mechanical removal, accumulating research suggests the procedure may trigger broader immunological shifts, potentially influencing T-cell populations and cytokine networks (Lehmann et al., Archives of Neurology, 2006).
Distinguishing Therapeutic from Donor Procedures
Two fundamentally different applications share the plasmapheresis name:
Therapeutic plasmapheresis aims to treat disease by removing harmful plasma constituents. The extracted plasma is discarded as medical waste.
Source plasma collection harvests plasma from healthy volunteers for pharmaceutical manufacturing - producing medications like immunoglobulin preparations and clotting factor concentrates.
This article addresses therapeutic applications exclusively.
Conditions Where Plasmapheresis May Provide Benefit
The American Society for Apheresis (ASFA) maintains regularly updated, evidence-graded recommendations for therapeutic apheresis applications. Their classification system assigns each indication to one of four categories reflecting the strength of supporting evidence (Connelly-Smith et al., Journal of Clinical Apheresis, 2023):
Category I: Accepted as first-line treatment, either independently or alongside other therapies
- Category II: Generally accepted as supportive or second-line treatment
- Category III: Optimal role remains undefined; individual assessment required
- Category IV: Published evidence demonstrates lack of efficacy or potential harm
The 2023 ASFA guidelines encompass 166 separately graded disease indications, with therapeutic plasma exchange appearing as a recommended modality in 118 of these entries.
Neurological Applications
Disorders affecting peripheral and central nervous system function represent some of the best-established plasmapheresis indications:
Guillain-Barré syndrome carries ASFA's highest recommendation (Category I, Grade 1A). This acute inflammatory polyneuropathy causes rapidly progressive weakness when misdirected immune responses attack peripheral nerve components. The landmark Cochrane analysis pooling six randomized trials demonstrated that plasma exchange significantly reduced mechanical ventilation requirements and accelerated functional recovery compared to supportive management alone (Chevret et al., Cochrane Database of Systematic Reviews, 2017). Additional details appear in our companion article examining plasmapheresis specifically for Guillain-Barré syndrome.
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) represents the chronic counterpart to GBS. The American Academy of Neurology assigns plasmapheresis Level A status (established effectiveness) for short-term CIDP management, with Cochrane data showing substantial improvement in approximately 80% of treated individuals (Mehndiratta et al., Cochrane Database of Systematic Reviews, 2019; Cortese et al., Neurology, 2011).
Myasthenia gravis responds to plasmapheresis during crisis episodes and before thymectomy surgery. Comparative systematic reviews suggest plasma exchange and intravenous immunoglobulin achieve similar overall outcomes, though some evidence points toward more rapid initial improvement with plasmapheresis (Kumar et al., Frontiers in Neurology, 2021).
Acute multiple sclerosis exacerbations that fail corticosteroid therapy may warrant plasmapheresis consideration (AAN Level B recommendation).
Hematological Disorders
Thrombotic thrombocytopenic purpura (TTP) presents perhaps the most compelling case for plasmapheresis efficacy. Before effective treatment existed, this condition carried roughly 90% mortality. Introduction of plasma exchange combined with immunosuppression has transformed outcomes dramatically—current survival rates now exceed 80% (Scully et al., British Journal of Haematology, 2012).
Hyperviscosity syndromes, where abnormal protein accumulation thickens blood dangerously, may benefit from plasma volume reduction.
Autoimmune and Inflammatory Conditions
Anti-glomerular basement membrane disease (historically called Goodpasture syndrome) holds Category I status for removing the pathogenic antibodies attacking kidney and lung tissue.
ANCA-associated vasculitis with severe manifestations - particularly pulmonary hemorrhage or rapidly declining kidney function—may warrant adjunctive plasmapheresis.
Transplantation Support
Plasma exchange facilitates ABO-incompatible organ transplantation by reducing recipient antibody titers and helps manage antibody-mediated rejection episodes in transplant recipients.
Understanding the Benefits of Plasmapheresis
Evaluating plasmapheresis benefits requires examining condition-specific evidence rather than making broad generalizations.
Direct Pathogen Removal
The procedure's fundamental mechanism involves physically extracting disease-driving substances from circulation. Each session processes approximately 1 to 1.5 total plasma volumes, achieving substantial reduction in targeted molecules. Serial treatments provide cumulative clearance effects (Nguyen et al., 2023).
Quantified Clinical Outcomes
The Cochrane GBS analysis provides concrete outcome measurements (Chevret et al., 2017):
Ventilator dependence: 47% relative reduction (risk ratio 0.53, meaning treated patients were roughly half as likely to require breathing support)
- Walking recovery at one month: 60% relative improvement (risk ratio 1.60 for regaining assisted ambulation)
- Disability improvement: 64% greater likelihood of advancing at least one functional grade within four weeks
- Complete strength recovery at twelve months: 24% relative advantage over supportive care alone
For TTP, the transformation from near-certain fatality to greater than 80% survival represents one of medicine's more dramatic therapeutic successes.
Duration Considerations
How long benefits persist varies considerably across conditions:
Guillain-Barré syndrome: When treatment begins within the first week of motor symptom onset, improvements generally persist. Evidence supports benefit even when treatment starts up to 30 days after onset, though earlier intervention correlates with better outcomes (Chevret et al., 2017; French Cooperative Group, Annals of Neurology, 1997).
CIDP: Approximately two-thirds of initially responding patients experience symptom recurrence within one to two weeks after completing treatment, suggesting many will require ongoing maintenance exchanges or transition to alternative long-term therapies (Mehndiratta et al., 2019).
TTP: Roughly 40% of patients experience disease recurrence over time, necessitating continued surveillance and potential retreatment.
Myasthenia gravis: Benefits typically last weeks to several months; many patients require repeated courses or combination immunotherapy.
Second-line rescue response rates (when plasmapheresis follows unsuccessful corticosteroid treatment) range between 50% and 85% depending on the specific condition (Nguyen et al., 2023).
Side Effects and Safety Considerations
Like any medical intervention, plasmapheresis carries potential adverse effects that warrant informed discussion.
Commonly Encountered Effects
Systematic complication surveillance has documented the following occurrence frequencies (Mörtzell Henriksson et al., Intensive Care Medicine, 2013):
Blood pressure drops affect approximately 8.4% of sessions, typically responding to fluid adjustment or temporary procedure slowing
- Citrate-related symptoms arise from the anticoagulant used during processing, manifesting as perioral tingling, extremity numbness, or muscle twitching; calcium supplementation usually provides rapid relief
- Sensory disturbances (paresthesias) occur in roughly 1% of treatments
- Temperature-related symptoms including chills may accompany plasma exchange
- Post-procedure fatigue is commonly reported
Less Frequent but Serious Complications
More significant adverse events occur infrequently but deserve awareness:
Cardiac rhythm disturbances appear in approximately 3.5% of procedures
- Vascular access problems - particularly with central venous catheters - include thrombosis and line-associated infections; peripheral access carries lower complication rates
- Hypersensitivity reactions to replacement solutions occur rarely
- Severe events (including circulatory shock, sustained arrhythmias, or blood cell destruction) affect roughly 2% of procedures
Reassurance from Controlled Trials
Critically, the Cochrane GBS analysis found no statistically significant increase in serious adverse events when comparing plasma exchange to supportive care. Pooled data from three trials encompassing 556 patients showed equivalent serious infection rates between groups (risk ratio 0.91; 95% confidence interval 0.73-1.13), with no clear differences in cardiovascular complications (Chevret et al., 2017).
When performed at experienced facilities with appropriate monitoring protocols, most side effects of plasmapheresis prove mild and self-limiting.
The Treatment Experience
Knowing what to anticipate may help patients prepare mentally and practically.
Pre-Treatment Preparation
- Vascular access establishment: Depending on vein quality and anticipated treatment duration, access may utilize peripheral arm veins (similar to standard IV placement) or require temporary central venous catheter insertion
- Baseline laboratory assessment: Blood tests establish starting values for comparison
- Medication coordination: Some medications may require timing adjustments around sessions
- Hydration guidance: Adequate fluid intake before and after treatment is typically encouraged
During Each Session
Standard plasmapheresis sessions follow this general pattern:
Time commitment: Two to four hours per session
- Exchange volume: One to one-and-a-half times total plasma volume processed
- Typical course: Five to six sessions distributed over ten to fourteen days, though protocols vary by condition and response
- Continuous monitoring: Vital signs and symptom surveillance throughout
- Activity during treatment: Patients often read, use devices, watch media, or simply rest
ASFA recommendations emphasize individualizing session number and frequency based on disease severity and clinical trajectory (Connelly-Smith et al., 2023).
Post-Treatment Recovery
- Normal activities can generally resume shortly after each session concludes
- Maintaining good hydration supports recovery
- Specific activity restrictions depend on individual circumstances and access type
- Follow-up blood work may monitor treatment response
Comparing Treatment Approaches
Understanding how plasmapheresis relates to alternative treatments helps contextualize its role.
Plasmapheresis Versus Dialysis
Despite superficial similarities - both involve blood processing through external circuits - these procedures serve entirely different purposes:
Plasmapheresis targets plasma-borne disease mediators (antibodies, immune complexes, abnormal proteins) and treats autoimmune, neurological, and hematological conditions.
Dialysis removes metabolic waste products and excess fluid, compensating for kidney failure.
The two cannot substitute for each other.
Plasmapheresis Versus Intravenous Immunoglobulin
For several overlapping indications—particularly GBS and myasthenia gravis - both plasmapheresis and IVIG hold first-line status. Multiple comparative analyses have examined their relative merits (Kumar et al., 2021; Zhang et al., Journal of Clinical Neuroscience, 2024):
Overall effectiveness: Comparable clinical outcomes across studied populations
- Speed of improvement: Some evidence suggests plasmapheresis produces faster initial response, particularly within the first two weeks
- Practical considerations: IVIG administration is generally simpler, potentially shortening hospitalization
- Adverse event profiles: No significant overall safety differences between approaches
Selection between these options typically reflects availability, individual patient factors, specific contraindications, and institutional experience rather than clear superiority of either approach.
Frequently Asked Questions
What conditions does plasmapheresis treat?
ASFA guidelines encompass 166 disease indications where plasmapheresis may play a role. The strongest evidence supports its use for Guillain-Barré syndrome, thrombotic thrombocytopenic purpura, myasthenia gravis crisis, and CIDP management.
Does plasmapheresis cause pain?
The procedure itself produces minimal discomfort. Vascular access placement involves brief needle insertion comparable to blood donation. Citrate-related tingling sensations, when they occur, respond quickly to calcium supplementation.
How long do treatment sessions last?
Individual sessions typically require two to four hours. Complete treatment courses usually involve five to six sessions over approximately two weeks, though specific protocols vary by indication.
What side effects should I expect?
Most commonly reported effects include temporary blood pressure changes, tingling from citrate anticoagulant, and post-treatment fatigue. Serious complications remain uncommon—roughly 2% of procedures - and most symptoms resolve quickly.
How does plasmapheresis differ from dialysis?
These procedures address fundamentally different problems. Plasmapheresis removes disease-causing plasma components and treats autoimmune or hematological conditions. Dialysis eliminates metabolic waste and excess fluid in kidney failure. One cannot replace the other.
How long do treatment benefits persist?
Duration varies substantially by condition. GBS improvements generally persist with early treatment. CIDP patients often experience recurrence within two weeks, potentially requiring maintenance therapy. TTP carries approximately 40% long-term relapse risk.
Does the procedure compromise immune function?
Plasmapheresis temporarily reduces circulating antibody levels, including protective immunoglobulins. The immune system regenerates these over subsequent weeks. Infection monitoring remains part of standard post-treatment care.
How does plasmapheresis compare to IVIG treatment?
For many shared indications, both approaches demonstrate similar effectiveness. Plasmapheresis may produce faster initial improvement; IVIG offers simpler administration. Treatment selection depends on individual circumstances, availability, and clinical judgment.
Who qualifies for plasmapheresis treatment?
Appropriate candidates typically have conditions with Category I or II ASFA designations, have not responded adequately to standard therapies, or require rapid reduction of pathogenic plasma substances. Specialist evaluation determines individual suitability.
What questions should I discuss with my physician?
Key discussion points include: Does my condition have established evidence supporting plasmapheresis? What treatment protocol would you recommend? What specific risks apply to my situation? What alternatives exist? What realistic outcomes can I anticipate?
Summary Points
- Plasmapheresis mechanically removes disease-driving plasma components through either centrifugal or membrane-based separation
- ASFA guidelines recognize 166 potential applications, with strongest evidence supporting treatment of GBS, TTP, myasthenia gravis, and CIDP
- Cochrane systematic reviews and randomized trials provide quantified benefit evidence for multiple indications
- Common treatment effects—blood pressure changes, citrate symptoms, fatigue—are typically mild and transient; serious complications affect roughly 2% of procedures
- Standard courses involve five to six sessions over two weeks, with each session lasting two to four hours
- Benefit duration ranges from sustained (early-treated GBS) to requiring maintenance (CIDP) depending on the underlying condition
- For shared indications, plasmapheresis and IVIG demonstrate comparable overall effectiveness
For those seeking comprehensive information about therapeutic plasma exchange within an evidence-based care framework, Humanaut Health provides Therapeutic Plasma Exchange (TPE) services.
References
- Chevret S, Hughes RAC, Annane D. "Plasma exchange for Guillain-Barré syndrome." Cochrane Database of Systematic Reviews. 2017;2:CD001798. DOI
- Connelly-Smith L, Alquist CR, Aqui NA, et al. "Guidelines on the Use of Therapeutic Apheresis in Clinical Practice: The Ninth Special Issue." Journal of Clinical Apheresis. 2023;38(2):77-278. DOI
- Cortese I, Chaudhry V, So YT, et al. "Evidence-based guideline update: Plasmapheresis in neurologic disorders." Neurology. 2011;76(3):294-300. DOI
- French Cooperative Group on Plasma Exchange in Guillain-Barré Syndrome. "Appropriate number of plasma exchanges in Guillain-Barré syndrome." Annals of Neurology. 1997;41(3):298-306. DOI
- Kumar R, et al. "Therapeutic Plasma Exchange in Myasthenia Gravis: A Systematic Literature Review and Meta-Analysis of Comparative Evidence." Frontiers in Neurology. 2021;12:662856. PMC
- Lehmann HC, Hartung HP, Hetzel GR, et al. "Plasma exchange in neuroimmunological disorders: Part 1." Archives of Neurology. 2006;63(7):930-935. DOI
- Mehndiratta MM, Hughes RAC, Agarwal P. "Plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy." Cochrane Database of Systematic Reviews. 2019. PMC
- Mörtzell Henriksson M, et al. "Complications in patients treated with plasmapheresis in the intensive care unit." Intensive Care Medicine. 2013;39(5):896-899. PubMed
- Nguyen TC, Kiss JE, Goldman JR, Carcillo JA. "Therapeutic Plasma Exchange: Core Curriculum 2023." American Journal of Kidney Diseases. 2023;81(4):493-503. AJKD
- Padmanabhan A, et al. "Plasmapheresis." StatPearls [Internet]. 2024. NCBI
- Scully M, Hunt BJ, Benjamin S, et al. "Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura." British Journal of Haematology. 2012;158(3):323-335. NCBI
- Zhang Y, et al. "Assessing the comparative efficacy of plasmapheresis and IVIG in myasthenia gravis treatment." Journal of Clinical Neuroscience. 2024;121:46-54. PubMed
[killen]








