Plasmapheresis for Myasthenia Gravis: How It Works and Benefits
When myasthenia gravis symptoms worsen - whether gradually or in sudden crisis -finding a treatment that works quickly becomes urgent. Plasmapheresis for myasthenia gravis offers exactly that: a direct approach that removes the antibodies causing your symptoms, often producing improvement within days rather than weeks. For many MG patients, this treatment represents a turning point when other therapies haven't provided adequate relief.
This guide explains everything you need to know about plasmapheresis for myasthenia gravis, including how the treatment works specifically for MG, how quickly you can expect plasma exchange to work, when doctors recommend this therapy, and what the research shows about its effectiveness. Whether you're facing a myasthenic crisis, preparing for thymectomy surgery, or exploring options for managing difficult symptoms, understanding plasmapheresis in myasthenia gravis will help you make informed decisions about your care.
What Is Myasthenia Gravis and Why Does Plasmapheresis Help?
Myasthenia gravis is an autoimmune condition where your immune system produces antibodies that attack the communication points between nerves and muscles. Specifically, most MG patients have antibodies targeting acetylcholine receptors (AChR) - the proteins that receive signals telling muscles to contract. When these receptors are blocked or destroyed by antibodies, the message from nerve to muscle gets disrupted, causing the characteristic weakness and fatigue of MG.
Understanding why plasmapheresis is done for myasthenia gravis starts with this antibody problem. Unlike treatments that suppress your immune system's ability to make new antibodies, plasmapheresis directly removes the harmful AChR antibodies already circulating in your blood. Think of it as clearing the battlefield rather than just stopping reinforcements—the antibodies causing your symptoms right now are physically extracted.
This removal provides rapid relief because once the antibodies are gone, your acetylcholine receptors can function more normally again. Nerve signals reach muscles more effectively, and strength improves. The relationship between antibody levels and symptoms is direct enough that researchers can measure both declining antibody counts and improving muscle function after myasthenia gravis plasmapheresis treatment.
For MG patients, this mechanism makes plasmapheresis particularly valuable during acute situations where waiting for slower-acting treatments isn't an option.
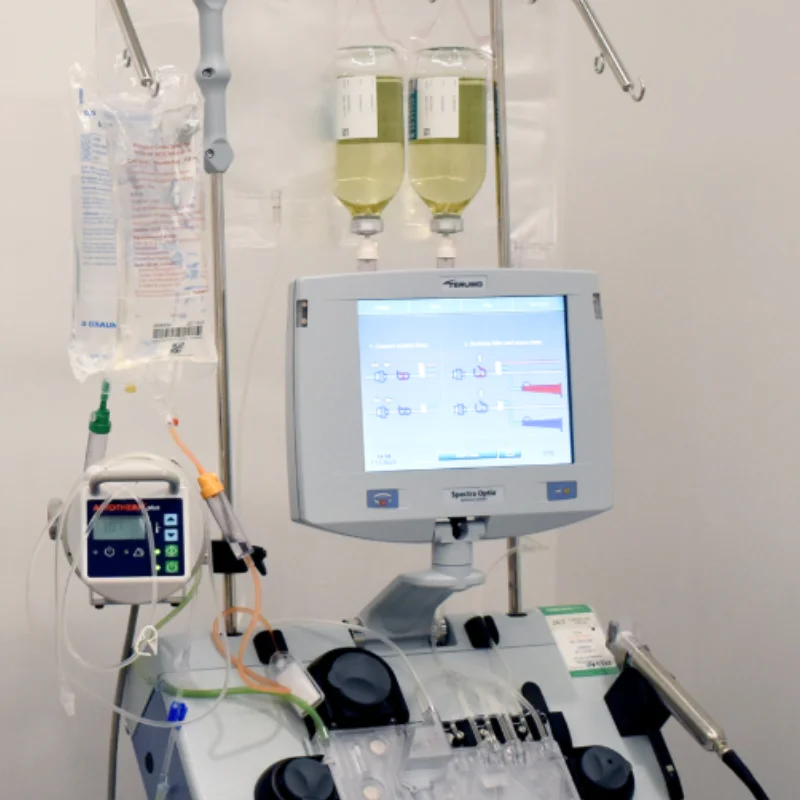
How Plasmapheresis Works for Myasthenia Gravis
The process of plasmapheresis for myasthenia gravis involves separating your blood into its components, removing the plasma containing harmful antibodies, and returning your blood cells with fresh replacement fluid. But what makes this especially effective for MG is the specific impact on the antibodies driving your disease.
Research shows that a single plasmapheresis session removes 60-71% of AChR antibodies from circulation (Guptill et al., 2016). Over a typical treatment course of multiple sessions, antibody levels drop dramatically - in some studies falling to just 21% of their original levels. This substantial reduction directly correlates with symptom improvement.
The antibody removal in myasthenia gravis and plasmapheresis works because IgG antibodies (the type that includes AChR antibodies) have a specific half-life in your body. Under normal circumstances, it takes about 21 days for half of these antibodies to naturally break down. Plasmapheresis accelerates this process dramatically, achieving in hours what would otherwise take weeks.
Importantly, plasmapheresis in myasthenia gravis doesn't eliminate your protective immunity. Studies confirm that antibodies against diseases like tetanus, diphtheria, and chickenpox remain at protective levels even after treatment. The procedure reduces all antibodies proportionally, but protective levels are maintained because you started with much higher concentrations than needed for immunity.
After plasmapheresis, your body will gradually produce new antibodies—including, unfortunately, new AChR antibodies. This is why plasmapheresis for myasthenia gravis is typically combined with immunosuppressive medications that slow new antibody production, extending the benefits of treatment.
How Quickly Does Plasma Exchange Work for MG?
One of the most common questions MG patients ask is how quickly does plasma exchange work? The answer is encouraging: most patients experience improvement within days to two weeks, making plasmapheresis one of the fastest-acting treatments available for myasthenia gravis.
Timeline of Improvement
First Few Days: Some patients notice changes after just one or two sessions. If you're in myasthenic crisis with breathing difficulties, improvement may be apparent within the first few treatments as antibody levels begin dropping.
Week 1-2: This is when most patients experience their most significant measurable improvements. Research shows that clinical response—defined as meaningful improvement on standardized MG assessment scales—typically occurs within 7-14 days of starting treatment (Cochrane Database, 2002).
Week 2-4: Maximum benefits are usually achieved during this period. Patients who respond to treatment generally reach their best improvement level within a month of completing their course.
What Improvement Looks Like
When we say plasmapheresis for myasthenia gravis works quickly, what does that mean practically? Patients typically experience:
Reduced double vision or drooping eyelids
- Improved swallowing ability
- Better breathing capacity
- Increased limb strength
- Less fatigue with activity
- Reduced need for rest breaks
In clinical terms, studies show a 46% average improvement in MG severity scores after plasmapheresis treatment (El Shakankiry et al., 2021). That translates to meaningful functional gains in daily life.
Crisis vs Non-Crisis Response
How quickly plasma exchange works can vary based on your situation. In myasthenic crisis—when weakness affects breathing muscles—improvement often comes faster because treatment is aggressive and closely monitored. Some crisis patients show measurable improvement within the first 24-48 hours.
For less acute exacerbations, the timeline may extend slightly, with gradual improvement building over the full treatment course. Either way, myasthenia gravis plasmapheresis typically produces faster results than waiting for immunosuppressive medications to take full effect.
When Is Plasmapheresis Recommended for Myasthenia Gravis?
Understanding why plasmapheresis is done for myasthenia gravis in different situations helps you understand when your doctor might recommend this treatment. The American Society for Apheresis (ASFA) classifies plasmapheresis for MG as a first-line or second-line therapy depending on the clinical scenario (ASFA Guidelines, 2023).
Myasthenic Crisis (ASFA Category I - First-Line)
Myasthenic crisis occurs when MG weakness affects the muscles controlling breathing, potentially requiring ventilator support. This is a medical emergency where plasmapheresis for myasthenia gravis serves as first-line treatment alongside respiratory support.
In crisis situations, the rapid antibody removal from plasmapheresis can be life-saving. Research shows that patients in crisis who receive myasthenia gravis plasmapheresis have shorter times on mechanical ventilation and faster recovery compared to other approaches.
Pre-Thymectomy Preparation
Many MG patients benefit from surgical removal of the thymus gland (thymectomy). Plasmapheresis in myasthenia gravis before surgery helps optimize your condition for the procedure. Studies demonstrate significant benefits:
Mechanical ventilation time: 1.8 hours with pre-operative TPE vs 2.9 hours without
- ICU stay: 1.4 days vs 2.6 days
- Reduced risk of post-operative myasthenic crisis
By reducing antibody levels before surgery, plasmapheresis for myasthenia gravis helps ensure smoother recovery and fewer complications.
Acute Exacerbations
When MG symptoms suddenly worsen - whether from infection, stress, medication changes, or other triggers - plasmapheresis provides rapid stabilization. This is particularly valuable when:
Symptoms are severe enough to affect daily function significantly
- First-line medications aren't controlling the flare adequately
- Rapid improvement is needed (upcoming surgery, important life events)
Refractory Disease
Some patients have MG that doesn't respond well to standard immunosuppressive medications. For these individuals, periodic myasthenia gravis and plasmapheresis may serve as maintenance therapy, with treatments scheduled regularly to keep symptoms controlled.
Plasmapheresis vs IVIG for Myasthenia Gravis
If your doctor is discussing acute MG treatment options, you've likely heard about both plasmapheresis and intravenous immunoglobulin (IVIG). Both are effective, but understanding their differences helps clarify why plasmapheresis for myasthenia gravis might be recommended for you.
Head-to-Head Comparison
A comprehensive meta-analysis comparing these treatments found that plasmapheresis achieves a 19% higher response rate than IVIG for acute myasthenia gravis (p=0.002) (Ipe et al., 2021). This means that in studies directly comparing the two, more patients improved with myasthenia gravis plasmapheresis than with IVIG.
When Plasmapheresis May Be Preferred
Faster onset: Plasmapheresis in myasthenia gravis typically works more quickly than IVIG, making it preferable when rapid improvement is critical.
Higher response rates: The research advantage of +19% means better odds of meaningful improvement.
When IVIG hasn't worked: Some patients who don't respond adequately to IVIG will respond to plasmapheresis for myasthenia gravis.
Severe or crisis situations: The speed and reliability of plasmapheresis make it the go-to choice for myasthenic crisis.
When IVIG May Be Preferred
Easier administration: IVIG doesn't require the specialized equipment plasmapheresis needs.
Vascular access: Patients with poor veins may tolerate IVIG better.
Outpatient convenience: IVIG can sometimes be given in settings where plasmapheresis isn't available.
Fewer immediate side effects: Some patients experience fewer reactions with IVIG.
Combination Approaches
In some situations, doctors use both treatments together or sequentially. Myasthenia gravis and plasmapheresis combined with IVIG or immunosuppressants may provide better long-term control than any single approach.
What to Expect: Treatment Protocol and Sessions
Knowing what plasmapheresis for myasthenia gravis involves practically helps you prepare and know what to expect.
Typical Treatment Course
Number of sessions: Most treatment courses involve 3-6 sessions. Patients in myasthenic crisis may need 5-6 or more sessions until stabilized.
Frequency: Sessions are typically scheduled every other day (alternate-day protocol), though this can be adjusted based on your response.
Duration: Each session takes approximately 2-4 hours.
Volume exchanged: Approximately one plasma volume (about 3-4 liters) is processed per session.
During Each Session
A typical myasthenia gravis plasmapheresis session involves:
Vascular access: Blood is drawn through an IV line (peripheral or central catheter depending on your situation)
- Separation: A machine separates your blood into plasma and cells
- Replacement: Your plasma is discarded and replaced with albumin solution
- Return: Your blood cells plus new fluid return to your body
- Monitoring: Staff check vital signs and watch for any reactions throughout
Many patients read, watch videos, or rest during treatment. You'll remain connected to the machine but can move somewhat and adjust position.
Preparing for Treatment
Before starting plasmapheresis in myasthenia gravis:
Blood tests will check your baseline levels
- Vascular access will be established
- Your medications may be adjusted (some are affected by plasmapheresis)
- You'll learn signs of reactions to watch for
After Treatment
Following each session, you may feel fatigued and should plan to rest. Between sessions, monitor for any unusual symptoms and report them to your care team. The full benefit of plasmapheresis for myasthenia gravis builds over the treatment course.
Benefits and Efficacy: What the Research Shows
The evidence supporting plasmapheresis for myasthenia gravis is substantial, making it one of the best-studied applications of this treatment.
Response Rates
Research consistently shows response rates of 50-100% for myasthenia gravis plasmapheresis, depending on the patient population and how response is measured (Frontiers in Neurology, 2021). In meta-analyses, all comparative studies showed response rates of at least 50%, with many reporting much higher success.
Measurable Improvements
Studies document specific improvements after plasmapheresis in myasthenia gravis:
MG severity scores: Average 46% improvement
- Antibody clearance: 56-71% reduction
- Ventilation time: Reduced from 2.9 to 1.8 hours (pre-surgery patients)
- ICU stay: Reduced from 2.6 to 1.4 days (pre-surgery patients)
- Functional improvement at 4 weeks: 57% of patients vs 35% without treatment
Quality of Life
Beyond clinical measurements, myasthenia gravis and plasmapheresis treatment shows meaningful quality of life benefits. Studies report:
78% of patients rate their treatment experience as good
- Reduced medication requirements after treatment
- Psychological and psychosocial improvements
- Better ability to perform daily activities
Duration of Benefits
The improvement from plasmapheresis for myasthenia gravis typically lasts 2-4 weeks after completing treatment. This is why most patients receive concurrent immunosuppressive therapy - to maintain benefits beyond the immediate antibody removal effect.
Without maintenance immunosuppression, antibody levels gradually rise again. Some patients experience "antibody overshoot," where levels actually exceed pre-treatment values around 6 weeks post-treatment. Proper combination therapy prevents this rebound.
Side Effects and Safety Considerations
Like any medical procedure, myasthenia gravis plasmapheresis carries some risks. Understanding these helps you recognize problems early and feel confident about treatment.
Common Side Effects (15-21% of patients)
Citrate reactions: The anticoagulant used during plasmapheresis can temporarily bind calcium, causing tingling around the mouth or fingers, and muscle cramps. This is easily treated with calcium supplementation during the procedure.
Hypotension: Some patients experience temporary low blood pressure, causing lightheadedness. Staff monitor this and can adjust the procedure if needed.
Fatigue: Feeling tired after sessions is common. Plan for rest on treatment days.
Less Common Reactions
Catheter-related issues: If a central line is used, there's small risk of infection, bleeding, or clotting at the site. Proper care minimizes these risks.
Allergic reactions: Rare, but can occur particularly if fresh frozen plasma is used as replacement fluid. Using albumin replacement reduces this risk.
The Antibody Rebound Phenomenon
One consideration specific to plasmapheresis in myasthenia gravis is potential antibody rebound. In some patients, antibody levels increase significantly—sometimes exceeding original levels - around 4-6 weeks after treatment ends (Muscle & Nerve, 2021).
This is why plasmapheresis for myasthenia gravis is almost always combined with immunosuppressive medications. The immunosuppression prevents the rebound by reducing new antibody production while the plasmapheresis handles existing antibodies.
Overall Safety Profile
Despite these considerations, myasthenia gravis plasmapheresis has a well-established safety record. Serious complications are rare, and most side effects are manageable. The benefits for appropriate patients—particularly those in crisis or with severe symptoms—clearly outweigh the risks, which is why major medical guidelines support its use.
Conclusion
Plasmapheresis for myasthenia gravis stands as one of the most effective and fastest-acting treatments available for this challenging condition. By directly removing the AChR antibodies causing your symptoms, this treatment offers something medications alone cannot: rapid improvement, often within days to two weeks.
The evidence is strong. Response rates reach 50-100% in studies. Myasthenia gravis plasmapheresis achieves 19% better outcomes than IVIG. Patients in crisis can stabilize quickly. Those preparing for surgery have fewer complications. And for those whose symptoms haven't responded to other treatments, plasmapheresis in myasthenia gravis offers a proven alternative.
If you're living with myasthenia gravis and facing worsening symptoms, myasthenic crisis, upcoming surgery, or inadequate response to current medications, plasmapheresis deserves serious consideration. The treatment's ability to produce meaningful improvement quickly makes it valuable in situations where waiting simply isn't acceptable.
At Humanaut Health, we provide therapeutic plasma exchange with the expertise and personalized attention this treatment requires. Our team understands the nuances of plasmapheresis for myasthenia gravis and works closely with patients to optimize outcomes. If you'd like to learn more about whether plasmapheresis might help your MG, we're here to discuss your options.
References
1. Guptill, J. T., et al. (2016). "Plasmapheresis for the treatment of myasthenia gravis: A systematic review." Neurology, 87(2), 243–251. https://doi.org/10.1212/WNL.0000000000002877








