Cholesterol and Plasmapheresis: How Plasma Exchange Lowers Lipids
For most people with high cholesterol, medications like statins and lifestyle changes are enough to bring lipid levels under control. But for those with familial hypercholesterolemia - a genetic condition that causes dangerously elevated LDL cholesterol from birth — even maximum-dose drug therapy may not be sufficient. This is where plasmapheresis for high cholesterol enters the picture. Also known as LDL apheresis or lipoprotein apheresis, this FDA-approved procedure physically removes LDL cholesterol and other harmful lipoproteins from the blood, reducing LDL levels by up to 85 percent in a single session. Studies suggest it may reduce cardiovascular events by 50 to 85 percent in eligible patients, yet it remains dramatically underused - with fewer than 400 of an estimated 11,000 to 15,000 eligible Americans currently receiving treatment.
[banner2]
This guide explains how plasmapheresis for high cholesterol works, who qualifies, what the evidence shows, and why lipoprotein(a) reduction is an emerging opportunity in preventive cardiovascular care.
Understanding Familial Hypercholesterolemia
Familial hypercholesterolemia (FH) is one of the most common inherited genetic disorders, affecting approximately 1 in 313 people worldwide - roughly 31 million individuals globally (Hu et al., JACC, 2020). The more severe form, homozygous FH (HoFH), occurs in approximately 1 in 300,000 people.
FH is caused by mutations in genes that regulate LDL receptor function, most commonly the LDLR gene. These mutations impair the body's ability to clear LDL cholesterol from the bloodstream, leading to severely elevated LDL levels from childhood. Without treatment, individuals with HoFH may develop coronary artery disease in their teens or twenties. Even heterozygous FH (HeFH) carries a significantly elevated cardiovascular risk — FH is 10 times more prevalent among patients with ischemic heart disease and 20 times more prevalent among those with premature heart attacks.
Despite these risks, fewer than 10 percent of FH patients worldwide have been diagnosed. The condition often goes unrecognized until a cardiovascular event occurs — a preventable tragedy for a treatable condition.
For patients with HoFH, statins and other cholesterol-lowering drugs depend on functioning LDL receptors to work effectively. When those receptors are severely impaired or absent, medications alone may not adequately reduce LDL. This is where apheresis becomes essential - it removes LDL physically, bypassing the receptor pathway entirely (Cuchel et al., European Heart Journal, 2023).
How Plasmapheresis for High Cholesterol Works
Plasmapheresis in the context of cholesterol management refers to a family of extracorporeal procedures that selectively remove atherogenic lipoproteins from the blood. While standard therapeutic plasma exchange (TPE) removes all plasma components non-selectively, LDL apheresis for familial hypercholesterolemia uses specialized technology to target and remove specific harmful particles while preserving beneficial components like HDL cholesterol.
Several LDL apheresis methods are available:
Dextran sulfate adsorption - uses negatively charged beads to bind and trap positively charged LDL particles
- Heparin-induced extracorporeal LDL precipitation (HELP) - precipitates LDL using heparin at acidic pH
- Immunoadsorption - uses antibodies to selectively capture apolipoprotein B-containing lipoproteins
- Direct adsorption of lipoproteins (DALI)- whole-blood system that removes LDL without separating plasma
All of these methods achieve a similar result: the rapid, dramatic removal of LDL cholesterol, lipoprotein(a), VLDL, and other atherogenic particles from the circulation. A single session typically takes 2 to 4 hours, and the reduction in LDL is immediate - unlike medications that require weeks to months to achieve full effect.
Importantly, LDL apheresis works independently of LDL receptor function. This makes it effective even in patients with receptor-negative HoFH, where statins and PCSK9 inhibitors have limited or no effect.
ASFA Guidelines and FDA Approval
Plasmapheresis for high cholesterol is both guideline-supported and FDA-approved — yet remains one of the most underutilized evidence-based therapies in cardiovascular medicine.
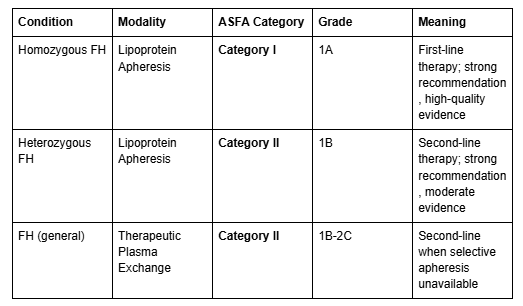
ASFA Classification
The American Society for Apheresis (ASFA) 2023 guidelines provide clear classifications for lipid apheresis (Connelly-Smith et al., Journal of Clinical Apheresis, 2023):
FDA-Approved Indications
LDL apheresis has been FDA-approved since 1996 (Liposorber LA-15 System). Patients must meet one of four eligibility groups:
Group A: HoFH with LDL-C >500 mg/dL
- Group B: HeFH with LDL-C ≥300 mg/dL despite maximally tolerated drug therapy
- Group C: HeFH with LDL-C ≥200 mg/dL + documented coronary artery disease
- Group D: Elevated Lp(a) ≥60 mg/dL + LDL-C ≥100 mg/dL + progressive cardiovascular disease (added in 2018 via Humanitarian Device Exemption)
A Dramatically Underused Therapy
The 2024 American Heart Association Scientific Statement described lipoprotein apheresis as "valuable but underused," noting that only approximately 400 of an estimated 11,000 to 15,000 eligible Americans currently receive treatment. About 60 apheresis centers exist across the United States (Gianos et al., Arteriosclerosis, Thrombosis, and Vascular Biology, 2024).
The European Atherosclerosis Society (EAS) 2023 consensus update recommends initiating lipoprotein apheresis by age 5 in children with HoFH, and no later than age 8, to prevent irreversible cardiovascular damage.
What the Evidence Shows
LDL Cholesterol Reduction
The cholesterol-lowering effect of apheresis is dramatic. A systematic review of 38 studies found the following per-session reductions (Moriarty et al., JAHA, 2016):
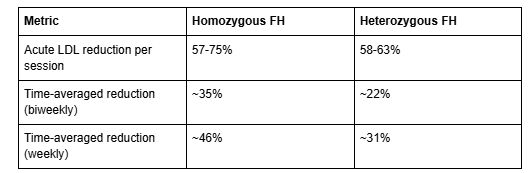
When weekly apheresis is combined with multidrug therapy, time-averaged LDL-C reductions of 70 to 80 percent compared to untreated baseline have been reported. The AHA Scientific Statement notes that individual sessions may reduce LDL-C by up to 85 percent.
Cardiovascular Outcome Reduction
The impact on cardiovascular events is equally compelling:
Mabuchi 1998 (Hokuriku Study): In a 6-year study of 130 heterozygous FH patients, the coronary event rate was 72 percent lower in the LDL apheresis group (10%) compared to drug therapy alone (36%) (p=0.0088) (Mabuchi et al., American Journal of Cardiology, 1998)
Leebmann 2013 (Pro(a)LiFe Study): In 170 patients with Lp(a)-hyperlipoproteinemia and progressive CVD, the mean annual rate of major adverse cardiovascular events decreased from 0.41 to 0.09 — a 78 percent reduction (Leebmann et al., Circulation, 2013)
Roeseler 2015: Across 36,745 lipoprotein apheresis treatments in 118 patients, cardiovascular events were reduced by 80 percent over a 6-year period (Roeseler et al., Atherosclerosis Supplements, 2015)
AHA 2024: Observational studies collectively suggest a 50 to 85 percent reduction in major adverse cardiovascular events
Landmark Randomized Trials
Two early randomized trials established the foundation for lipid apheresis:
- Thompson 1995 (FHRS): The first major RCT in heterozygous FH demonstrated that fortnightly LDL apheresis combined with simvastatin was more effective than medication alone at reducing both LDL-C and Lp(a) (Thompson et al., The Lancet, 1995)
- Kroon 1996 (LAARS): A randomized study of 42 patients showed that biweekly apheresis achieved a 63 percent LDL reduction (vs. 47% with medication alone), arrested coronary disease progression in both groups, and produced a 39 percent improvement in exercise capacity in the apheresis group (Kroon et al., Circulation, 1996)
The Lipoprotein(a) Opportunity
One of the most significant emerging applications of apheresis is the reduction of lipoprotein(a), or Lp(a) — a genetically determined lipoprotein that is an independent risk factor for cardiovascular disease and cannot be meaningfully lowered by diet, exercise, or most medications.
Lipoprotein(a) apheresis treatment is particularly relevant for individuals focused on longevity and cardiovascular risk optimization, as elevated Lp(a) affects approximately 20 percent of the population and is associated with accelerated atherosclerosis.
Key facts about Lp(a) and apheresis:
- A single apheresis session reduces Lp(a) by 60 to 75 percent
- The Pro(a)LiFe study demonstrated a 78 percent reduction in cardiovascular events in patients with Lp(a)-hyperlipoproteinemia
- LDL apheresis is currently the only FDA-indicated treatment that significantly lowers Lp(a) — PCSK9 inhibitors reduce Lp(a) by only approximately 25 to 30 percent by comparison
- FDA Group D specifically covers patients with elevated Lp(a) (≥60 mg/dL) plus LDL-C ≥100 mg/dL plus progressive cardiovascular disease
As awareness of Lp(a) as a cardiovascular risk factor grows — and as novel Lp(a)-lowering drugs continue through clinical trials — lipoprotein(a) apheresis treatment represents an important bridge therapy for high-risk patients.
How Apheresis Compares to Modern Medications
LDL apheresis is not a replacement for medication — it is typically used alongside pharmacotherapy for patients who cannot achieve adequate lipid control with drugs alone. Here is how the approaches compare:
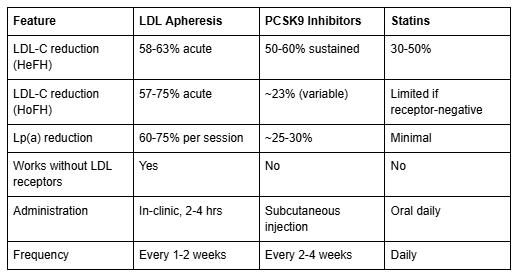
The key advantage of apheresis is that it works independently of LDL receptor function — making it the only effective option for patients with receptor-negative HoFH. For most patients with HeFH, apheresis is added when maximally tolerated pharmacotherapy (statins + ezetimibe + PCSK9 inhibitors) fails to achieve target LDL levels.
Novel therapies such as evinacumab (an angiopoietin-like 3 inhibitor) and lomitapide (a microsomal triglyceride transfer protein inhibitor) may reduce the frequency of apheresis sessions for some HoFH patients, but apheresis remains a cornerstone of treatment.
Safety and Side Effects
LDL apheresis has an excellent long-term safety profile — some patients have been receiving regular treatments for decades.
In a study of 36,745 lipoprotein apheresis treatments, adverse events occurred in only 1.1 percent of sessions. No serious complications were reported. The most common side effects included temporary hypotension (0.8%), mild nausea (0.5%), and flushing (0.4%).
Important safety considerations:
- Patients taking ACE inhibitors must switch to ARBs (angiotensin receptor blockers) before treatment with dextran sulfate apheresis systems, due to the risk of bradykinin-mediated reactions
- Temporary fatigue and lightheadedness may occur during or after sessions
- Catheter-related complications (bruising, infection) are possible but uncommon
- LDL apheresis has a significantly lower adverse event rate (approximately 2.9%) compared to standard therapeutic plasma exchange (approximately 12%)
The procedure is well-tolerated enough that many patients continue biweekly treatments for years or even decades as part of their ongoing cardiovascular risk management.
What to Expect During Treatment
LDL apheresis is performed as an outpatient procedure, typically in a specialized apheresis center or hospital clinic.
Treatment protocol:
- Frequency: Every 1 to 2 weeks, depending on the severity of hypercholesterolemia and treatment response
- Duration: Each session takes approximately 2 to 4 hours
- Access: Blood is drawn through a peripheral IV or central venous catheter, processed through the apheresis device, and returned to the patient
- Monitoring: Lipid panels, Lp(a) levels, blood counts, and coagulation parameters are checked regularly
What patients should understand:
- LDL levels rebound between sessions — this is expected and is why regular treatments are necessary
- The procedure treats the symptom (elevated LDL) but not the genetic cause — it is a lifelong management strategy for most FH patients
- Patients typically continue taking cholesterol-lowering medications alongside apheresis
- Improvement in cardiovascular risk markers is typically evident within the first few months of regular treatment
Frequently Asked Questions
Can plasmapheresis cure high cholesterol?
No. Plasmapheresis for high cholesterol is a management strategy, not a cure. It physically removes LDL and other harmful lipoproteins from the blood, but levels rebound between sessions because the underlying genetic cause (in FH) remains. Regular treatments are needed to maintain lower lipid levels.
How much does LDL apheresis reduce cholesterol?
A single session of LDL apheresis for familial hypercholesterolemia typically reduces LDL cholesterol by 57 to 85 percent. However, because LDL rebounds between sessions, the time-averaged reduction with biweekly treatment is approximately 22 to 35 percent, depending on the form of FH. Weekly sessions combined with medication may achieve 70 to 80 percent sustained reduction.
Who qualifies for LDL apheresis?
FDA-approved indications include: (A) homozygous FH with LDL >500 mg/dL; (B) heterozygous FH with LDL ≥300 mg/dL despite maximal drug therapy; (C) heterozygous FH with LDL ≥200 mg/dL plus documented coronary disease; and (D) elevated Lp(a) ≥60 mg/dL with LDL ≥100 mg/dL and progressive cardiovascular disease. All patients must be on maximally tolerated lipid-lowering therapy.
Is plasmapheresis for high cholesterol FDA-approved?
Yes. LDL apheresis has been FDA-approved since 1996 (Liposorber LA-15 System). It is also recommended by the ASFA as Category I (first-line) for homozygous FH and Category II (second-line) for heterozygous FH, and endorsed by the American Heart Association, European Atherosclerosis Society, and other major medical organizations.
How often do you need LDL apheresis?
Most patients receive treatments every 1 to 2 weeks. The frequency depends on the severity of hypercholesterolemia, treatment response, and whether the patient has homozygous or heterozygous FH. Each session takes approximately 2 to 4 hours.
Does LDL apheresis lower Lp(a)?
Yes. Lipoprotein(a) apheresis treatment reduces Lp(a) by 60 to 75 percent per session — significantly more than any currently available medication. PCSK9 inhibitors, by comparison, reduce Lp(a) by only approximately 25 to 30 percent. Apheresis is currently the only FDA-indicated treatment for clinically significant Lp(a) reduction.
What are the side effects of LDL apheresis?
LDL apheresis has an excellent safety profile. In a study of over 36,000 treatments, adverse events occurred in only 1.1 percent of sessions. The most common side effects are temporary hypotension, mild nausea, and flushing. Patients taking ACE inhibitors must switch to ARBs before treatment due to the risk of bradykinin reactions with certain apheresis systems.
How does LDL apheresis compare to PCSK9 inhibitors?
Both are effective for lowering LDL cholesterol, but they work differently. PCSK9 inhibitors (evolocumab, alirocumab) achieve 50 to 60 percent sustained LDL reduction in HeFH but only about 23 percent in HoFH, because they depend on LDL receptor activity. LDL apheresis achieves 57 to 85 percent acute reduction per session and works regardless of receptor function. For Lp(a), apheresis achieves 60 to 75 percent reduction versus 25 to 30 percent with PCSK9 inhibitors.
Key Takeaways
- Plasmapheresis for high cholesterol (LDL apheresis) is an FDA-approved, guideline-supported treatment that reduces LDL by 57 to 85 percent per session
- It is classified as ASFA Category I (first-line) for homozygous FH and Category II (second-line) for heterozygous FH
- Studies show 50 to 85 percent reduction in major cardiovascular events — including a 72 percent reduction in the landmark Mabuchi study
- LDL apheresis for familial hypercholesterolemia works independently of LDL receptor function, making it effective even when drugs cannot achieve adequate control
- Lipoprotein(a) apheresis treatment reduces Lp(a) by 60 to 75 percent per session — currently the only FDA-indicated approach for significant Lp(a) lowering
- The procedure is safe: adverse events in only 1.1 percent of over 36,000 treatments, with no serious complications reported
- Despite strong evidence, it remains dramatically underused — only about 400 of 15,000 eligible Americans currently receive treatment
Take the Next Step
If you are living with familial hypercholesterolemia, elevated Lp(a), or treatment-resistant high cholesterol, Humanaut Health offers therapeutic plasma exchange as part of a personalized, evidence-based approach to cardiovascular health and longevity optimization. Contact our team to discuss whether lipid apheresis may be appropriate for your situation.
References
- Connelly-Smith L, Alquist CR, Aqui NA, et al. "Guidelines on the Use of Therapeutic Apheresis in Clinical Practice — Evidence-Based Approach from the Writing Committee of the American Society for Apheresis: The Ninth Special Issue." Journal of Clinical Apheresis. 2023;38(2):77-278. DOI: 10.1002/jca.22043
- Gianos E, Sperling LS, et al. "Lipoprotein Apheresis: Utility, Outcomes, and Implementation in Clinical Practice: A Scientific Statement From the American Heart Association." Arteriosclerosis, Thrombosis, and Vascular Biology. 2024. DOI: 10.1161/ATV.0000000000000177
- Moriarty PM, Hemphill LC, Ballantyne CM, et al. "Lipoprotein Apheresis Efficacy, Outcomes, and Costs." Journal of the American Heart Association. 2016;5:e003294. DOI: 10.1161/JAHA.116.003294
- Mabuchi H, Koizumi J, Shimizu M, Takeda R; Hokuriku-FH-LDL-Apheresis Study Group. American Journal of Cardiology. 1998;82(12):1489-1495. PMID: 9874053
- Leebmann J, Roeseler E, Julius U, et al. "Lipoprotein Apheresis in Patients With Maximally Tolerated Lipid-Lowering Therapy, Lipoprotein(a)-Hyperlipoproteinemia, and Progressive Cardiovascular Disease." Circulation. 2013;128(24):2567-2576. DOI: 10.1161/CIRCULATIONAHA.113.002432
- Roeseler E, Julius U, Heigl F, et al. "Lipoprotein Apheresis for Lipoprotein(a)-Associated Cardiovascular Disease." Atherosclerosis Supplements. 2015;18:154-162. DOI: 10.1016/j.atherosclerosissup.2015.02.013
- Hu P, Dharmayat KI, Stevens CAT, et al. "Prevalence of Familial Hypercholesterolemia Among the General Population and Patients With Atherosclerotic Cardiovascular Disease." JACC. 2020;75(20):2553-2566. DOI: 10.1016/j.jacc.2020.03.057
- Cuchel M, Bruckert E, Ginsberg HN, et al. "2023 Update on European Atherosclerosis Society Consensus Statement on Homozygous Familial Hypercholesterolaemia." European Heart Journal. 2023;44(25):2277-2291. DOI: 10.1093/eurheartj/ehad197
- Thompson GR, Maher VMG, Matthews S, et al. "Familial Hypercholesterolaemia Regression Study." The Lancet. 1995;345(8953):811-816. PMID: 7898227
- Kroon AA, Aengevaeren WRM, van der Werf T, et al. "LDL-Apheresis Atherosclerosis Regression Study (LAARS)." Circulation. 1996;93(10):1826-1835. DOI: 10.1161/01.CIR.93.10.1826
[killen]








