Plasmapheresis Treatment for Lupus: How It Works and Benefits
Systemic lupus erythematosus doesn't attack just one organ - it can target the brain, lungs, blood vessels, kidneys, and skin simultaneously, often unpredictably. When lupus flares become life-threatening and standard immunosuppressive therapy isn't controlling the damage fast enough, plasmapheresis treatment for lupus offers a way to rapidly remove the harmful antibodies fueling the attack. For specific severe manifestations - catastrophic antiphospholipid syndrome, thrombotic thrombocytopenic purpura, and neuropsychiatric lupus among them - plasma exchange may be the difference between organ failure and recovery.
But plasmapheresis is not equally effective across all lupus complications. The evidence clearly distinguishes where it works from where it doesn't, and understanding that distinction is essential for patients and caregivers weighing treatment options. This guide examines the research behind plasmapheresis treatment for lupus, covering which manifestations respond best, what clinical outcomes look like, treatment protocols, safety considerations, and when this therapy is — and isn't — the right approach.
[banner2]
How Lupus Challenges the Immune System
Lupus is an autoimmune disease in which the immune system loses its ability to distinguish the body's own tissues from foreign threats. Instead of protecting you, the immune system produces autoantibodies - including anti-double-stranded DNA (anti-dsDNA), antiphospholipid, and anti-Smith antibodies — that attack healthy cells and tissues throughout the body.
These autoantibodies don't act alone. They form immune complexes that circulate in the blood, deposit in organs, and activate complement — a cascade of inflammatory proteins that amplifies tissue damage. This process can simultaneously affect the kidneys, brain, lungs, blood vessels, joints, and skin, making lupus one of the most complex autoimmune conditions to manage.
Standard treatment relies on immunosuppressive medications — corticosteroids, hydroxychloroquine, mycophenolate, cyclophosphamide, and biologics like belimumab — that reduce immune system activity over time. But these drugs take weeks to months to reach full effectiveness. During severe flares that threaten organ function or life, that timeline may not be fast enough.
How Plasmapheresis Works for Lupus
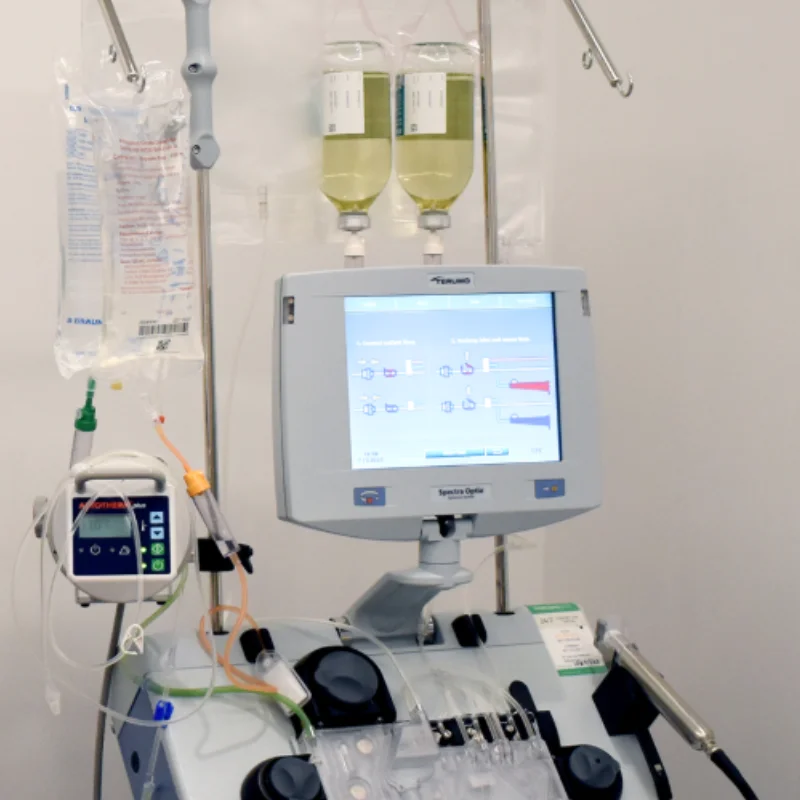
Plasmapheresis addresses the gap between a severe flare and the time immunosuppressive medications need to work. The procedure separates blood into its components, removes the plasma containing harmful autoantibodies and immune complexes, and returns blood cells with clean replacement fluid.
For lupus specifically, plasmapheresis targets several pathological components simultaneously:
- Autoantibodies (anti-dsDNA, antiphospholipid, anti-neuronal) that drive organ damage
- Circulating immune complexes that deposit in tissues and trigger inflammation
- Complement components (C3, C5) that amplify the inflammatory cascade
- Inflammatory cytokines that sustain tissue injury
- Cryoglobulins — cold-sensitive antibody complexes found in some lupus patients
The speed advantage matters in emergencies. While immunosuppressants may take weeks to reduce antibody production, a course of plasma exchange for lupus flares can lower circulating autoantibody levels by over 60% within the first few sessions. This rapid reduction creates a critical window for organs to begin recovering while long-term medications build to therapeutic levels.
Where Plasmapheresis Is Most Effective in Lupus
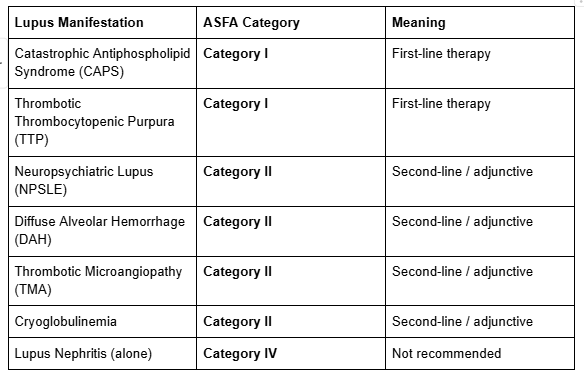
The American Society for Apheresis (ASFA) classifies therapeutic apheresis indications by the strength of evidence. For lupus, the classifications vary dramatically depending on which organ system is involved (Connelly-Smith et al., J Clin Apher, 2023):
Catastrophic Antiphospholipid Syndrome (CAPS) - First-Line, Life-Saving
CAPS is among the most dangerous complications of lupus. It involves widespread blood clotting in small vessels across multiple organs simultaneously — kidneys, lungs, brain, heart — and can progress to multi-organ failure within days. Though rare, CAPS carries a historically high mortality rate of 37-50%.
Plasmapheresis is part of the standard triple therapy for CAPS: anticoagulation, high-dose corticosteroids, and therapeutic plasma exchange. This combination achieves the highest survival rates documented — approximately 72% survival — by rapidly removing the antiphospholipid antibodies driving the clotting cascade while simultaneously addressing coagulation and inflammation (CAPS Registry data).
ASFA classifies CAPS as Category I, making plasmapheresis a first-line treatment. For patients with lupus who develop this catastrophic complication, plasma exchange is not optional — it is a core component of the most effective treatment protocol available.
Thrombotic Thrombocytopenic Purpura (TTP) - Category I, Strongest Evidence
TTP occurs when antibodies against the enzyme ADAMTS13 lead to uncontrolled blood clotting in small vessels, consuming platelets and causing organ damage. Lupus-associated TTP is a distinct complication that requires immediate treatment.
The evidence for plasmapheresis in TTP carries the highest ASFA recommendation - Category I, Grade 1A. Before plasma exchange became available, TTP had a mortality rate exceeding 90%. With plasmapheresis as the cornerstone of treatment, mortality has been reduced to approximately 5-10%, representing one of the most dramatic treatment advances in hematology.
Plasmapheresis works in TTP through a dual mechanism: it removes the harmful anti-ADAMTS13 antibodies and simultaneously supplies functional ADAMTS13 enzyme through the replacement plasma. Treatment typically involves daily sessions until platelet counts normalize and hemolysis resolves.
Neuropsychiatric Lupus (NPSLE) - Evidence of Significant Benefit
When lupus attacks the central nervous system, it can cause seizures, psychosis, stroke-like episodes, severe cognitive dysfunction, and coma. Neuropsychiatric lupus is classified as ASFA Category II supported as adjunctive therapy, particularly when patients are refractory to standard immunosuppression.
The evidence from case series is encouraging. In a review of 26 patients with severe CNS lupus treated with plasmapheresis, 74% improved, 13% stabilized, and only 13% progressed (Neuwelt, Ther Apher Dial, 2003). Notably, 11 of these patients were under 21 years old, underscoring the value of plasma exchange for lupus flares affecting the brain in younger patients who may not tolerate aggressive immunosuppression.
A separate study of 10 patients with 13 NPSLE flares found even stronger results: all patients improved within a mean of three weeks, with 54% achieving complete remission and 46% partial remission. Disease activity scores dropped dramatically — from 6.9 to 1.2 on the European Consensus Lupus Activity Measurement scale (Bartolucci et al., Lupus, 2007).
Diffuse Alveolar Hemorrhage (DAH) - Rescue Therapy
DAH - bleeding into the lung tissue - is a rare but potentially fatal lupus complication with historical mortality rates as high as 85%. Current treatment has improved survival to approximately 60-70%, but DAH remains a medical emergency.
Plasmapheresis is used as rescue therapy in approximately 31% of severe DAH cases when standard immunosuppressive treatment proves insufficient. Among those treated with plasma exchange, approximately 55% showed improvement (Aguirre-Valencia et al., Autoimmune Diseases, 2019). ASFA classifies DAH in lupus as Category II, supporting its use as adjunctive therapy in severe or refractory cases.
Thrombotic Microangiopathy (TMA) - The Important Exception
Here is a critical nuance for patients with lupus-related kidney disease. While standard lupus nephritis does not benefit from plasmapheresis (see next section), lupus nephritis that is accompanied by thrombotic microangiopathy -a distinct complication involving blood clot formation in small kidney vessels - does appear to benefit.
A study of 70 patients with biopsy-proven TMA in lupus nephritis found that those who received plasmapheresis in addition to standard therapy had significantly higher remission rates (P=0.018) and significantly fewer composite endpoints including kidney failure and death (P=0.005) compared to patients receiving standard therapy alone (Li et al., Medicine, 2016).
This distinction matters because it highlights a pattern: plasmapheresis for lupus is most effective when the damage is driven by circulating factors — antibodies, immune complexes, clotting abnormalities — that the procedure can physically remove.
When Plasmapheresis Is Not Recommended for Lupus
Despite lupus being an autoimmune disease with circulating autoantibodies, plasmapheresis is not recommended for lupus nephritis as a standalone indication. ASFA classifies it as Category IV — meaning evidence shows it does not provide benefit for routine use.
The evidence comes from a landmark multicenter randomized controlled trial of 86 patients with severe lupus nephritis. Adding plasmapheresis to standard immunosuppressive therapy (prednisone plus cyclophosphamide) provided no clinical benefit — kidney failure rates and mortality were not improved despite faster reduction of anti-dsDNA antibodies (Lewis et al., N Engl J Med, 1992). The trial was terminated early for futility.
Why does plasmapheresis fail in lupus nephritis but succeed in other lupus complications? The likely explanation lies in the mechanism of kidney damage. In lupus nephritis, much of the injury comes from immune complexes that have already deposited within the kidney tissue and from in-situ immune complex formation — processes that removing circulating antibodies cannot reverse. In contrast, complications like CAPS, TTP, and NPSLE are driven primarily by circulating factors that plasmapheresis can directly remove.
For a deeper discussion of plasmapheresis in kidney disease, including the conditions where it is effective (anti-GBM disease, ANCA vasculitis), see our dedicated guide.
The critical takeaway: lupus nephritis is not the same as lupus. A patient with lupus who also has CAPS, TTP, NPSLE, or DAH may absolutely benefit from plasmapheresis — even if their kidneys are also affected.
What the Research Shows
The largest real-world study of plasmapheresis treatment for lupus evaluated 66 patients with severe autoimmune disease, of whom 40 had SLE. The most common indications were diffuse alveolar hemorrhage (27.5%), neuropsychiatric lupus (22.5%), thrombotic microangiopathy (12.5%), and CAPS (10%). Overall, 62% of patients showed improvement, and importantly, there were no TPE-related deaths (Aguirre-Valencia et al., Autoimmune Diseases, 2019).
A systematic review examining plasma exchange and immunoadsorption in SLE and antiphospholipid syndrome confirmed that both techniques have favorable safety profiles and are beneficial for refractory disease, patients with contraindications to standard immunosuppression, and during pregnancy (Kronbichler et al., Autoimmun Rev, 2016).
Emerging evidence also suggests a role for plasmapheresis in managing lupus during pregnancy, where many immunosuppressive medications are contraindicated. A 2023 study found that plasma exchange improved key clinical markers — ESR, urinary protein, and platelet counts — in pregnant SLE patients and may help prevent antiphospholipid-related pregnancy complications (Zhang et al., Immunity Inflamm Dis, 2023).
It is worth noting that large randomized controlled trials for plasmapheresis in CAPS, TTP, and NPSLE are limited — not because the treatment is unproven, but because these are rare, life-threatening emergencies where randomization to a no-treatment arm would be ethically difficult. The consistent positive outcomes across multiple case series, registries, and expert consensus support its use in these settings.
Safety and Side Effects
Understanding plasmapheresis for lupus side effects helps patients make informed decisions. The procedure has a well-established safety profile across thousands of treatments for various conditions.
Common side effects (generally mild and temporary):
- Catheter site discomfort or bruising
- Temporary blood pressure fluctuations (hypotension in ~1-2%, hypertension in ~2%)
- Fatigue during or after sessions
- Chills or tingling (related to citrate anticoagulant)
- Mild allergic reactions to replacement fluid
Serious complications are uncommon, occurring in fewer than 3% of sessions. Major side effects are typically catheter-related — infection at the insertion site, bleeding — rather than from the plasma exchange procedure itself.
In the Aguirre-Valencia 2019 study of 66 patients, no deaths were attributed to the plasma exchange procedure. The main complication noted was hemorrhage, which was managed with appropriate interventions.
For lupus patients specifically, one additional consideration is infection risk. Plasmapheresis temporarily removes immunoglobulins (protective antibodies) along with the harmful autoantibodies. Since lupus patients are often already immunocompromised from disease activity and immunosuppressive medications, your medical team monitors closely for signs of infection during treatment.
What to Expect During Treatment
Plasma exchange for lupus flares follows condition-specific protocols, but the general experience is similar across indications.
CAPS: Daily or every-other-day sessions until clinical stabilization. May require 5-10 sessions depending on response. Fresh frozen plasma is typically used as replacement fluid (provides clotting factors).
TTP: Daily sessions until platelet counts normalize and hemolysis markers improve. Treatment may continue for several days beyond normalization to prevent relapse.
NPSLE: Typically 3-7 sessions over 1-2 weeks, adjusted based on neurological response. Often synchronized with cyclophosphamide pulses for combined effect.
DAH: Emergency protocol — sessions may begin immediately upon diagnosis, with frequency adjusted based on clinical response.
Each session lasts approximately 2-4 hours. Your medical team monitors vital signs throughout and checks blood counts, organ function markers, and antibody levels between sessions. Plasmapheresis treatment for lupus is almost always combined with immunosuppressive therapy - the plasma exchange handles existing harmful antibodies while medications prevent new ones from being produced.
Frequently Asked Questions
Can plasmapheresis cure lupus?
No. Plasmapheresis is not a cure for lupus — it is a treatment that may help manage severe flares by rapidly removing harmful autoantibodies and immune complexes from the blood. Lupus is a chronic condition requiring ongoing management. Plasmapheresis is used alongside immunosuppressive medications to control life-threatening complications.
Which types of lupus respond to plasmapheresis?
Plasmapheresis is most effective for lupus-related catastrophic antiphospholipid syndrome (CAPS), thrombotic thrombocytopenic purpura (TTP), neuropsychiatric lupus (NPSLE), diffuse alveolar hemorrhage (DAH), and thrombotic microangiopathy. It is not recommended for lupus nephritis as a standalone indication (Connelly-Smith et al., J Clin Apher, 2023).
Is plasmapheresis recommended for lupus nephritis?
Standard lupus nephritis is classified as ASFA Category IV — meaning plasmapheresis is not recommended based on a randomized trial showing no benefit (Lewis et al., N Engl J Med, 1992). However, lupus nephritis accompanied by thrombotic microangiopathy may benefit, as this involves a distinct mechanism that plasmapheresis can address.
How many sessions are needed for lupus flares?
The number of sessions depends on the specific complication and severity. NPSLE typically requires 3-7 sessions over 1-2 weeks. CAPS and TTP may require daily sessions until clinical markers stabilize — often 5-10 sessions or more. Your rheumatologist or hematologist adjusts the protocol based on your response.
What are the side effects of plasmapheresis for lupus?
Common plasmapheresis for lupus side effects include temporary fatigue, blood pressure changes, catheter site discomfort, and chills. Serious complications occur in fewer than 3% of sessions. No TPE-related deaths were reported in the largest real-world study of plasma exchange for SLE (Aguirre-Valencia et al., Autoimmune Diseases, 2019).
Can plasmapheresis be used during lupus pregnancy?
Emerging evidence suggests plasmapheresis may be a valuable option for managing lupus during pregnancy, particularly when many immunosuppressive medications are contraindicated. A 2023 study reported improvements in clinical markers and potential prevention of antiphospholipid-related pregnancy complications (Zhang et al., Immunity Inflamm Dis, 2023). Decisions should be made with a multidisciplinary team.
How does plasmapheresis compare to other lupus treatments?
Plasmapheresis is not a replacement for standard lupus therapy — it is an adjunctive treatment for severe or refractory manifestations. Standard care (corticosteroids, hydroxychloroquine, mycophenolate, rituximab) remains the foundation. Plasmapheresis adds rapid antibody removal when the situation is urgent and standard medications need time to work.
Is plasmapheresis covered by insurance for lupus?
Coverage varies by insurance plan and the specific indication. Plasmapheresis for ASFA Category I indications (CAPS, TTP) is generally covered as a medically necessary procedure. Coverage for Category II indications may require prior authorization. Discuss coverage with your insurance provider and treatment team before beginning therapy.

Key Takeaways
- Plasmapheresis treatment for lupus is highly effective for specific severe manifestations but not recommended for all lupus complications
- CAPS (Category I): Triple therapy including plasmapheresis achieves approximately 72% survival in a condition with historically 50% mortality
- TTP (Category I): Plasmapheresis reduced mortality from over 90% to under 10% — one of the most significant treatment advances in hematology
- NPSLE (Category II): 74-100% of patients improve with plasmapheresis in published case series
- Lupus nephritis alone (Category IV): Not recommended based on randomized trial evidence — but the TMA subgroup is an exception
- Safety profile is well-established: no TPE-related deaths in the largest real-world study, with serious complications in fewer than 3% of sessions
- Plasma exchange for lupus flares works as bridge therapy — rapidly removing harmful antibodies while immunosuppressive medications take effect
- Not all lupus is the same — the specific manifestation determines whether plasmapheresis is appropriate, making specialist evaluation essential
For patients with lupus exploring evidence-based therapies for severe or refractory disease, Humanaut Health offers therapeutic plasma exchange delivered with concierge-level, personalized care. If you'd like to learn more about whether plasmapheresis may fit into your treatment plan, our team is here to help.
References
- Connelly-Smith L, et al. "Guidelines on the Use of Therapeutic Apheresis in Clinical Practice — The Ninth Special Issue." J Clin Apher. 2023;38(2):77-278. DOI: 10.1002/jca.22043\
- Lewis EJ, Hunsicker LG, Lan SP, et al. "A Controlled Trial of Plasmapheresis Therapy in Severe Lupus Nephritis." N Engl J Med. 1992;326(21):1373-1379. DOI: 10.1056/NEJM199205213262101
- Neuwelt CM. "The Role of Plasmapheresis in the Treatment of Severe Central Nervous System Neuropsychiatric Systemic Lupus Erythematosus." Ther Apher Dial. 2003;7(2):173-182. PMID: 12918940
- Bartolucci P, Brechignac S, Cohen P, Le Guern V, Guillevin L. "Adjunctive Plasma Exchanges to Treat Neuropsychiatric Lupus: A Retrospective Study on 10 Patients." Lupus. 2007;16(4):254-262. DOI: 10.1177/0961203307081840
- Li QY, Yu F, Zhou FD, Zhao MH. "Plasmapheresis Is Associated With Better Renal Outcomes in Lupus Nephritis Patients With Thrombotic Microangiopathy: A Case Series Study." Medicine. 2016;95(18):e3595. DOI: 10.1097/MD.0000000000003595
- Aguirre-Valencia D, Posso-Osorio I, Bravo JC, et al. "Therapeutic Plasma Exchange as Management of Complicated Systemic Lupus Erythematosus and Other Autoimmune Diseases." Autoimmune Diseases. 2019;2019:5350960. DOI: 10.1155/2019/5350960
- Zhang J, et al. "Clinical Efficacy of Plasma Exchange in Systemic Lupus Erythematosus During Pregnancy." Immunity Inflamm Dis. 2023;11(9):e1041. DOI: 10.1002/iid3.1041
- Kronbichler A, Brezina B, Geetha D, et al. "Efficacy of Plasma Exchange and Immunoadsorption in Systemic Lupus Erythematosus and Antiphospholipid Syndrome: A Systematic Review." Autoimmun Rev. 2016;15(1):38-49. DOI: 10.1016/j.autrev.2015.08.010
- Ozguler Y, et al. "Plasma Exchange Therapy in Systemic Lupus Erythematosus: A Single-Center Retrospective Cohort Study." J Turk Soc Rheumatol. 2023;15(2):89-97. DOI: raed.galenos.2023.29591
- Pagnoux C, Korach JM, Guillevin L. "Indications for Plasma Exchange in Systemic Lupus Erythematosus in 2005." Lupus. 2005;14(11):871-877. PMID: 16335578
[jeffrey]








