How Plasmapheresis Treats Multiple Sclerosis: Risks and Effectiveness
When a multiple sclerosis relapse strikes hard and intravenous steroids fail to bring relief, the question becomes: what comes next? For an estimated 20 to 40 percent of MS patients whose acute relapses do not respond adequately to first-line corticosteroid therapy, plasmapheresis for multiple sclerosis offers an evidence-based second-line treatment option. Also known as therapeutic plasma exchange (TPE), this procedure has been studied for over two decades in the context of MS, with response rates ranging from 40 to 79 percent across multiple clinical studies. This article explores how plasmapheresis works for MS, what the research shows about its effectiveness, who is most likely to benefit, and what risks to consider.
[banner2]
How Multiple Sclerosis Challenges the Immune System
Multiple sclerosis is a chronic autoimmune disease in which the immune system mistakenly attacks the protective myelin sheath surrounding nerve fibers in the brain and spinal cord. This process, known as demyelination, disrupts the transmission of nerve signals and may cause symptoms ranging from numbness and vision problems to weakness, difficulty walking, and cognitive changes.
During an MS relapse - also called an exacerbation or flare - the immune system launches an acute inflammatory attack against the central nervous system. This involves autoantibodies, complement proteins, and inflammatory cytokines that cross the blood-brain barrier and damage myelin. The standard first-line treatment for acute relapses is high-dose intravenous methylprednisolone (IV steroids), which helps reduce inflammation in most cases.
However, research indicates that a significant proportion of MS patients — approximately 20 to 40 percent — do not respond adequately to IV steroids during a severe relapse. For these individuals, the relapse may continue to cause progressive neurological damage without additional intervention.
How Plasmapheresis for Multiple Sclerosis Works
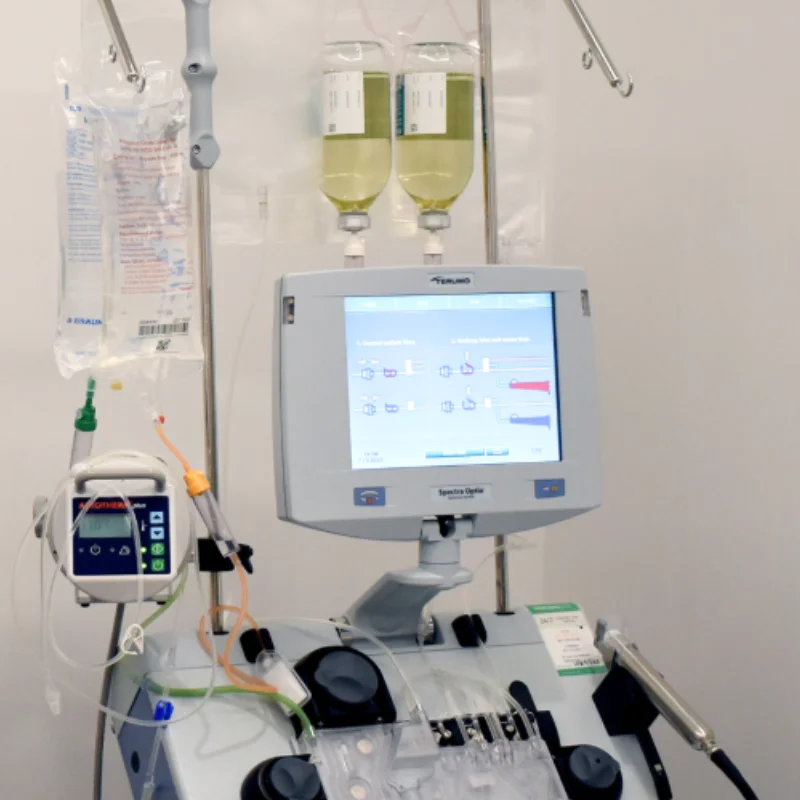
Plasmapheresis is a blood-filtering procedure that separates plasma — the liquid portion of blood containing antibodies, complement proteins, and inflammatory mediators — from blood cells. The plasma is removed and replaced with a substitute solution (typically albumin), and the blood cells are returned to the patient.
In the context of MS, plasmapheresis works by rapidly removing circulating autoantibodies, immune complexes, complement components, and pro-inflammatory cytokines that are driving the acute inflammatory attack. Unlike steroids, which suppress the immune system broadly and take days to weeks to achieve full effect, plasma exchange for MS relapse provides a more direct and rapid approach to clearing the specific inflammatory substances from the bloodstream.
This speed is a key advantage. By quickly reducing the concentration of harmful immune factors, plasmapheresis may help limit ongoing myelin damage and create a window for neurological recovery. It is typically used as a bridge therapy — buying time for the acute inflammation to subside while longer-term disease-modifying treatments continue to work.
ASFA Guidelines: When Plasmapheresis Is Recommended for MS
The American Society for Apheresis (ASFA) publishes evidence-based guidelines that classify medical conditions into categories based on how well therapeutic apheresis is supported by clinical evidence. The most recent edition — the 2023 Ninth Special Issue — includes specific recommendations for MS (Connelly-Smith et al., Journal of Clinical Apheresis, 2023).
ASFA Classification for Multiple Sclerosis

The critical distinction: Plasmapheresis is supported as a second-line treatment specifically for acute MS relapses that do not respond to IV corticosteroids. It is not recommended as a treatment for chronic progressive or secondary progressive MS. The American Academy of Neurology (AAN) classifies plasmapheresis as Level A evidence — established as ineffective — for chronic progressive forms of MS.
This distinction matters because MS involves both acute inflammatory attacks (relapses) and gradual neurodegeneration (progression). Plasmapheresis addresses the acute inflammatory component but does not appear to alter the long-term disease course.
A related condition worth noting is neuromyelitis optica spectrum disorder (NMOSD), which shares some clinical features with MS but involves different immune mechanisms. NMOSD acute attacks are also classified as ASFA Category II for plasmapheresis treatment, reflecting a similar role as second-line therapy.
What the Evidence Shows
The Landmark Trial: Weinshenker 1999
The foundational evidence for plasmapheresis in MS comes from a randomized, sham-controlled, double-masked crossover trial conducted by Weinshenker and colleagues at the Mayo Clinic. This Class I study enrolled patients with severe neurological deficits from CNS inflammatory demyelinating disease who had failed IV corticosteroids (Weinshenker et al., Annals of Neurology, 1999).
The results were striking: moderate or greater improvement occurred during 42.1 percent of active plasma exchange courses compared to just 5.9 percent of sham treatments. Improvement appeared early in treatment and was sustained on follow-up. This trial remains the highest-quality evidence supporting plasma exchange for MS.
Response Rates Across Multiple Studies
Since the Weinshenker trial, numerous studies have evaluated plasmapheresis for steroid-refractory MS relapses. The response rates vary depending on patient selection criteria and how "response" is defined, but the overall pattern is consistent:
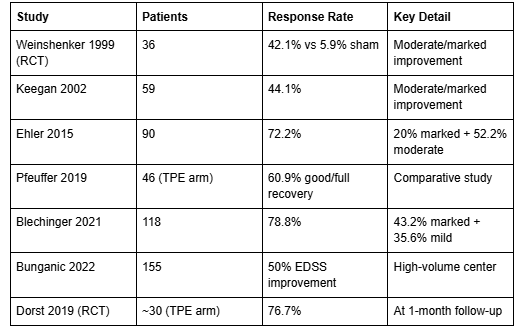
In the Ehler 2015 study of 90 patients, 72.2 percent showed a clinical response to plasma exchange, with median disability scores (EDSS) improving significantly from 3.75 to 3.0 (p=0.001) (Ehler et al., PLOS ONE, 2015). A two-center study by Blechinger and colleagues found that 78.8 percent of 118 MS patients improved following TPE — including 43.2 percent with marked improvement (Blechinger et al., Therapeutic Advances in Neurological Disorders, 2021). The Dorst 2019 randomized trial confirmed a 76.7 percent response rate at one month (Dorst et al., eClinicalMedicine, 2019).
How Plasmapheresis Compares to Escalated Steroids
One of the most compelling studies comes from Pfeuffer and colleagues, who compared plasma exchange directly to escalated IV methylprednisolone in 145 patients with steroid-refractory MS relapses (Pfeuffer et al., Journal of Clinical Medicine, 2019).
The results strongly favored plasmapheresis:
- TPE group: 60.9% achieved good or full recovery; only 6.5% had worst or no recovery
- Escalated steroids group: 15.2% achieved good or full recovery; 69.7% had worst or no recovery
- The odds ratio for worst/no recovery favoring TPE was 39.01 (95% CI: 10.41-146.18; p≤0.001)
Additionally, 53.5 percent of patients who initially received escalated steroids subsequently required TPE rescue — and 54.8 percent of those patients then responded satisfactorily.
When Improvement Begins
Research by Magana and colleagues suggests that patients who respond to plasmapheresis tend to show improvement early — within a median of 4 days, typically by the third treatment session. A small minority (approximately 6 percent) may show delayed improvement after two or more months (Magana et al., Archives of Neurology, 2011).
Who Benefits Most from Plasmapheresis for MS
Not every MS patient responds equally to plasmapheresis. Research has identified several factors associated with better outcomes:
Active inflammation is the strongest predictor. Patients with gadolinium-enhancing lesions on MRI — indicating active, ongoing inflammation — are significantly more likely to respond. In the Blechinger 2021 study, gadolinium enhancement was associated with an odds ratio of 3.2 for improvement. Ehler 2015 found this was the single best predictor of response (p=0.001).
Relapsing forms of MS respond better than progressive forms. Patients with relapsing-remitting MS (RRMS) or clinically isolated syndrome (CIS) showed significantly better responses than those with progressive disease (OR=3.1 in Blechinger 2021; p=0.001 in Ehler 2015).
Earlier treatment initiation is associated with better outcomes. Each 7-day delay in starting TPE was associated with reduced odds of improvement (OR=0.7 per week of delay). Research suggests that the greatest benefit may occur when plasmapheresis is initiated within 14 days of symptom onset.
Additional factors associated with better response include younger age, lower disability scores before treatment (EDSS ≤5.0; p=0.014), and preserved reflexes (Keegan et al., Neurology, 2002). Ring-enhancing lesions and mass effect on MRI have also been linked to favorable outcomes (Magana et al., Archives of Neurology, 2011).
In summary: active inflammation combined with early intervention appears to offer the best chance of meaningful improvement.
Plasmapheresis MS Side Effects and Safety
Understanding the risks associated with any medical procedure is essential for informed decision-making. The safety profile of plasmapheresis for multiple sclerosis has been well-characterized across multiple studies.
The overall picture is reassuring. In a high-volume center analysis of 603 TPE procedures in 155 MS patients, 94 percent of procedures had zero adverse events. Adverse events occurred in only 6 percent of procedures and were predominantly mild (Bunganic et al., Scientific Reports, 2022).
Serious complications are uncommon. Moser and colleagues reported a serious complication rate of 1.8 percent across 219 procedures, including one case of subclavian vein thrombosis, one episode of bradycardia with syncope, and two cases of impaired coagulation. Critically, zero deaths were reported across all major MS-specific TPE studies (Moser et al., Brain Sciences, 2019).
Common plasmapheresis MS side effects include:
- Catheter-site issues (3.7% of procedures)
- Vasovagal reactions — lightheadedness, fainting (1.8%)
- Allergic reactions (0.5%)
- Temporary drops in blood pressure (hypotension)
- Fatigue during or after sessions
- Mild chills or tingling
Potential but less common complications include:
- Catheter-related infection
- Deep venous thrombosis
- Temporary anemia
- Low albumin levels (hypoalbuminemia)
- Bleeding or coagulation changes
For MS patients, there is an additional consideration: many individuals are already taking immunosuppressive disease-modifying therapies, which may increase the risk of infection. Healthcare providers typically monitor patients closely and may adjust immunosuppressive medications during the treatment course.
What to Expect During Treatment
The ASFA-recommended protocol for plasma exchange for MS relapse involves 5 to 7 treatment sessions over a period of 10 to 14 days. Sessions are typically performed daily or every other day.
During each session:
- The procedure takes approximately 2 to 4 hours
- A volume of 1 to 1.5 times the patient's total plasma volume is exchanged per session
- Blood is drawn through a catheter, plasma is separated and removed, and blood cells are returned with replacement fluid
- Patients are monitored for vital signs and any adverse reactions throughout
What happens after treatment:
- Improvement may begin as early as the third session (median day 4), though some patients require the full treatment course
- Plasmapheresis treats the acute relapse — it is not a disease-modifying therapy and does not prevent future relapses
- Patients typically continue their baseline disease-modifying therapy for long-term MS management
- Follow-up monitoring includes neurological assessments, blood counts, and imaging as indicated
At 6-month follow-up in the Blechinger 2021 study, 27 percent of patients experienced new relapses — reinforcing that TPE addresses the acute inflammatory event while long-term disease management requires ongoing therapy.
Frequently Asked Questions
Is plasmapheresis a cure for multiple sclerosis?
No. Plasmapheresis is not a cure for MS. It is a treatment for acute MS relapses that do not respond to IV corticosteroids. It works by removing inflammatory substances from the blood during an active flare but does not alter the underlying disease course. Patients continue to need disease-modifying therapy for long-term management.
How effective is plasmapheresis for MS relapses?
Studies show response rates ranging from approximately 40 to 79 percent in patients with steroid-refractory MS relapses. The landmark Weinshenker trial found 42.1 percent moderate or marked improvement with active treatment versus 5.9 percent with sham. More recent studies report higher rates — up to 78.8 percent — likely reflecting improved patient selection and earlier treatment initiation.
When is plasmapheresis recommended for MS?
Plasmapheresis for multiple sclerosis is recommended as a second-line treatment (ASFA Category II, Grade 1B) specifically for acute relapses that do not respond adequately to high-dose IV corticosteroids. It is not recommended for chronic progressive or secondary progressive MS.
Does plasmapheresis work for progressive MS?
Current evidence does not support the use of plasmapheresis for chronic progressive or secondary progressive MS. The ASFA classifies this as Category III (role not established), and the AAN has classified it as Level A evidence — established as ineffective — for progressive forms of MS. Plasmapheresis targets acute inflammation, which is the hallmark of relapses rather than the gradual neurodegeneration seen in progressive MS.
How many plasmapheresis sessions are needed for MS?
The ASFA recommends 5 to 7 treatment sessions over 10 to 14 days. Sessions are typically performed daily or every other day. Each session lasts approximately 2 to 4 hours. Some patients may show improvement as early as the third session.
What are the side effects of plasmapheresis for MS?
The most common plasmapheresis MS side effects include catheter-site issues, vasovagal reactions (lightheadedness), mild allergic reactions, temporary blood pressure changes, and fatigue. In large studies, 94 percent of procedures had no adverse events. Serious complications — such as deep venous thrombosis or infection — occurred in approximately 1.8 percent of procedures. No deaths were reported in major MS-specific studies.
How quickly does plasmapheresis work for MS relapses?
Research suggests that patients who respond to plasmapheresis typically show improvement within a median of 4 days — by the third treatment session. However, a small percentage of patients (approximately 6 percent) may experience delayed improvement over two or more months. Earlier initiation of treatment is associated with better outcomes.
Is plasmapheresis covered by insurance for MS?
Coverage varies by insurance provider and individual plan. Because plasmapheresis for multiple sclerosis relapses is supported by ASFA Category II guidelines and AAN recommendations, many insurance plans do cover the procedure when medical necessity is documented. Patients should verify coverage details with their insurance provider before treatment.
Key Takeaways
- Plasmapheresis for multiple sclerosis is an evidence-based second-line treatment for acute relapses that do not respond to IV steroids, with response rates of 40 to 79 percent across multiple studies
- The landmark Weinshenker RCT showed 42.1 percent improvement with active treatment versus 5.9 percent with sham — the strongest evidence supporting this approach
- When compared directly to escalated steroids, plasmapheresis showed significantly better outcomes (60.9% vs 15.2% good recovery; OR=39.01)
- Active inflammation (gadolinium-enhancing MRI lesions) and early treatment initiation (within 14 days) are the strongest predictors of a positive response
- Plasmapheresis is not recommended for chronic progressive MS (ASFA Category III; AAN Level A — ineffective)
- The procedure has a favorable safety profile: 94 percent of procedures had zero adverse events, serious complications occurred in 1.8 percent of cases, and no deaths were reported in major studies
- Plasmapheresis treats the acute relapse but is not a disease-modifying therapy — ongoing MS management requires continued treatment
Take the Next Step
If you or a loved one is living with MS and exploring advanced treatment options, Humanaut Health offers therapeutic plasma exchange as part of a comprehensive, personalized approach to care. Our team works closely with each patient to determine whether TPE may be appropriate based on individual clinical circumstances.
References
- Weinshenker BG, O'Brien PC, Petterson TM, et al. "A randomized trial of plasma exchange in acute central nervous system inflammatory demyelinating disease." Annals of Neurology. 1999;46(6):878-886. DOI: 10.1002/1531-8249(199912)46:6<878::AID-ANA10>3.0.CO;2-Q
- Keegan M, Pineda AA, McClelland RL, et al. "Plasma exchange for severe attacks of CNS demyelination: predictors of response." Neurology. 2002;58(1):143-146. PMID: 11781423
- Ehler J, Blechinger S, Rommer PS, et al. "Response to Therapeutic Plasma Exchange as a Rescue Treatment in Clinically Isolated Syndromes and Acute Worsening of Multiple Sclerosis: A Retrospective Analysis of 90 Patients." PLOS ONE. 2015;10(8):e0134583. DOI: 10.1371/journal.pone.0134583
- Pfeuffer S, Rommer PS, Bsteh G, et al. "Comparing Plasma Exchange to Escalated Methyl Prednisolone in Refractory Multiple Sclerosis Relapses." Journal of Clinical Medicine. 2019. DOI: 10.3390/jcm9041077
- Blechinger S, Ehler J, Bsteh G, et al. "Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses: A retrospective two-center study." Therapeutic Advances in Neurological Disorders. 2021;14:1756286420975642. DOI: 10.1177/1756286420975642
- Bunganic R, et al. "Therapeutic plasma exchange in multiple sclerosis patients with an aggressive relapse: an observational analysis in a high-volume center." Scientific Reports. 2022;12:18535. DOI: 10.1038/s41598-022-23356-w
- Moser T, et al. "Therapeutic Plasma Exchange in Multiple Sclerosis and Autoimmune Encephalitis: A Comparative Study of Indication, Efficacy, and Safety." Brain Sciences. 2019;9(10):267. DOI: 10.3390/brainsci9100267
- Connelly-Smith L, et al. "Guidelines on the Use of Therapeutic Apheresis in Clinical Practice — Evidence-Based Approach from the Writing Committee of the American Society for Apheresis: The Ninth Special Issue." Journal of Clinical Apheresis. 2023;38(2):77-278. DOI: 10.1002/jca.22043
- Dorst J, Fangerau T, Taranu D, et al. "Safety and efficacy of immunoadsorption versus plasma exchange in steroid-refractory relapse of multiple sclerosis and clinically isolated syndrome: A randomised, parallel-group, controlled trial." eClinicalMedicine. 2019;16:98-106. DOI: 10.1016/j.eclinm.2019.10.004
- Magana SM, Keegan BM, Weinshenker BG, et al. "Beneficial plasma exchange response in central nervous system inflammatory demyelination." Archives of Neurology. 2011;68(7):870-878. DOI: 10.1001/archneurol.2011.34
[adelson]








